Edwards Lifesciences Corp., an Irvine-based medical device maker, said today that it received Food and Drug Administration clearance for its ClearSight device.
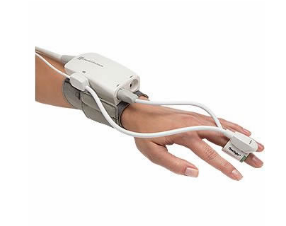
ClearSight is an advanced noninvasive patient monitor that provides access to blood volume and blood flow information for doctors. It is used on patients who are at moderate or high risk of post-surgery complications.
The device uses a cuff placed on the outside of a patient’s finger and is connected to Edwards’ EV1000 monitoring system.
Shares of Edwards were down 2% to a market value of $9.1 billion.