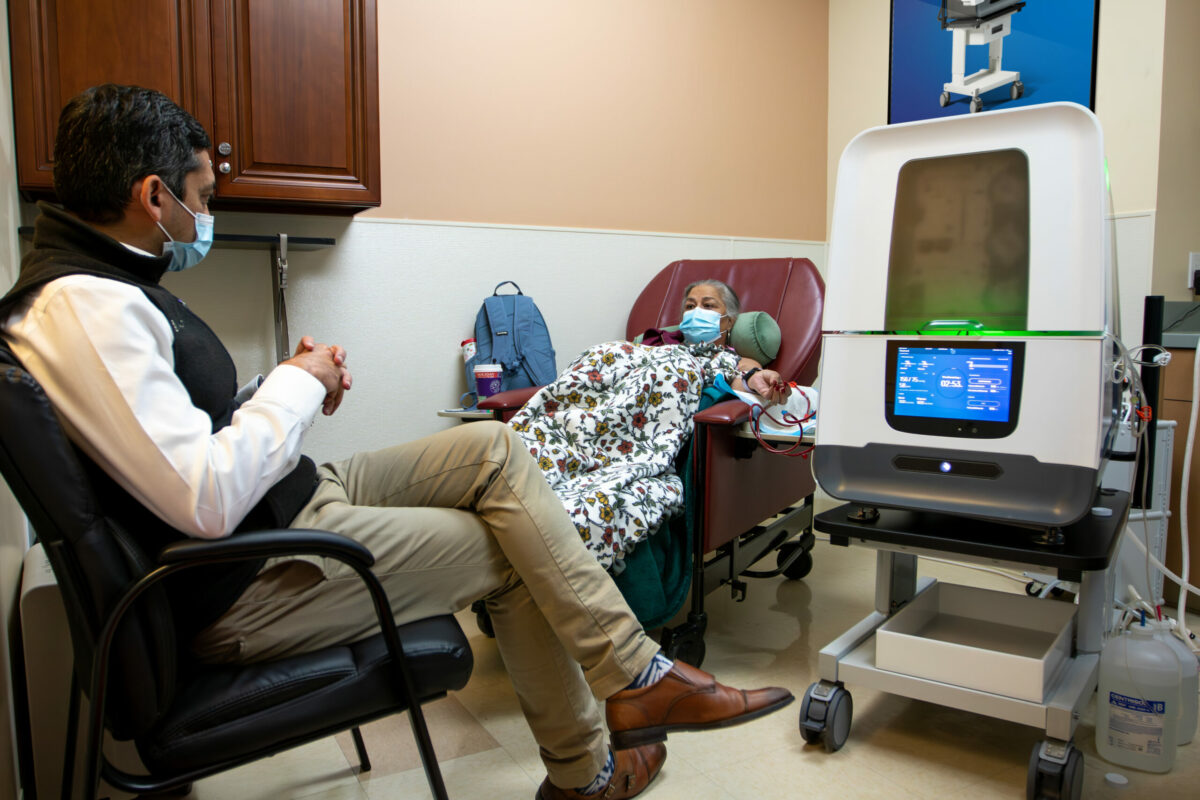
Diality Inc. has treated the first patient in a clinical trial for what it calls a paradigm-shifting kidney disease treatment.
The Irvine-based medical device company is partnering with aQua Research Institute and aQua Dialysis in Houston, Texas, on the trial to evaluate a home use indication for its Moda-flx Hemodialysis System.
“Getting the first patient treated in this home trial is another step in our journey to try and treat kidney disease,” Diality Chief Executive Osman Khawar told the Business Journal.
Receiving at-home use would give patients the freedom to receive treatment outside of clinics, where patients often spend hours.
The mobile system, which acts as an artificial kidney by filtering patients’ blood, received FDA clearance last August for use in professional care settings.
Entering Early Stages of Commercialization
Diality said it’s preparing for the early stages of commercialization of Moda-flx to targeted care settings in hospitals, dialysis units and skilled nursing facilities.
Last December, the company had its first post-approval activity. Its device was placed in select dialysis units for patient use. “We captured some data from that which is supporting our value proposition,” Khawar said.
The company submitted this data to the American Society of Nephrology and will be presenting it in November.
It has added seasoned executives with commercial experience to the board, including Dr. Chris Springer, whose addition was announced in March.
“Chris’s commercial experience and strategic focus is exactly what Diality needs as we transition to the early stages of commercialization of the Moda-flx Hemodialysis System,” Khawar said in a statement.
Springer is said to bring 30 years of marketing and strategy experience in the pharmaceutical and biotech spaces.
A large part of his career was spent as chief strategy officer of Swiss pharmaceutical company Vifor Pharma, where he oversaw licensing deals and the company’s acquisition for $11.7 billion by CLS Limited in 2022.
Springer is also on the boards of other healthcare companies including Adrenomed, Nagi Bioscience, Quercis Pharma and HICT.
$85B and Growing Market
The company, founded in 2018, has already raised more than $100 million to date.
Its most recent fundraising was in 2022 when it closed on a $24 million Series B2 round, which helped fund getting 510(k) clearance.
Nearly 555,000 people are on dialysis within the U.S., according to the American Kidney Fund.
The kidney disease treatment market was valued at $85 billion in 2024 and is projected to reach more than $120 billion by 2030.
Last month, Diality announced that it joined Kidney Care Partners, a nonprofit coalition of more than 30 organizations focused on advancing policies that benefits patients with kidney disease.