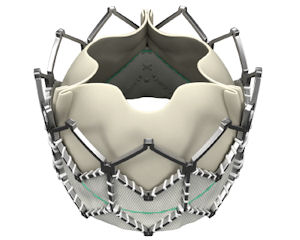
To get an idea of why investors and analysts are excited about Irvine-based Edwards Lifesciences Corp.’s latest heart valve, look to Europe.
The company’s Sapien valve, which is inserted via a catheter and doesn’t require major surgery, has been sold in Europe since 2007.
Since then, it has become the fastest-growing product for a company known for steady, predictable revenue gains.
Sapien, which could see U.S. approval next year or by 2012, is projected to see 2010 European sales of $190 million to $205 million, which would be up 70% to 80% from 2009.
Driving Growth
The valve still is a small part of Edwards, which has projected 2010 sales of $1.5 billion. But Sapien accounted for just about all of the company’s added revenue in the second quarter.
Edwards’ total sales grew by 9%, or by $30 million, to $365 million for the quarter. Sapien alone brought in $28 million in additional revenue in the quarter, offsetting slow-growing or declining products.
Sapien sales more than doubled to $53.2 million in the quarter.
The valve and competing devices are seen as the biggest advances in heart valves in years and could open up the market to millions of patients who aren’t candidates for getting their valves replaced via major surgery.
Analysts contend Sapien has the potential to double Edwards’ yearly sales in five years. It also could make the company a buyout target at a price of $8 billion to $10 billion, according to an analyst survey by Bloomberg.
Shares of Edwards, which are up more than 50% this year, had a recent market value of $7.5 billion.
Late last month, the New England Journal of Medicine published trial results from an initial U.S. study of Sapien that showed the valve significantly reduced death rates in patients implanted with it.
The trial covered patients who had aortic stenosis, or a narrowing of the valve that connects the heart to the body’s biggest artery.
The results were seen as an encouraging sign for eventual U.S. approval of Sapien. They also could boost already surging sales in Europe, according to one analyst.
“We also note that European market adoption of Sapien could accelerate due to the (trial) data,” said Spencer Nam, an analyst who follows Edwards for Madison Williams & Co., a New York investment bank.
The trial’s finding of lower death rates “alone could move several European countries to reimburse the procedures,” he said.
Nam said he’s “cautiously optimistic” that European Sapien sales could exceed $300 million in 2011.
“This clearly positive data likely will bode well for the future of the potentially $1 billion-plus” device, said Rick Wise of Boston-based Leerink Swann LLC in a report issued during a recent scientific meeting.
Edwards has an early lead in less-invasive valves but doesn’t have the market to itself.
Minnesota’s Medtronic Inc., which employs about 675 people in Irvine and Santa Ana, also sells less-invasive valves outside the U.S.
Medtronic is estimated to have half of the early market.
The company’s Irvine-based CoreValve unit, which Medtronic bought for $700 million in 2009, “is expected to enter a pivotal trial any day now,” Wise said in his note.
Edwards is on track to submit a pre-market approval application for Sapien in the fourth quarter, according to the company.
The true test for Edwards will come with a clinical trial showing how Sapien patients compare to those undergoing “higher-risk surgery,” said Duane Nash, an analyst with Los Angeles-based Wedbush Securities, in a research note.
Concerns
There are concerns with Sapien. The valve’s first trial showed a risk of strokes and vascular complications.
The stroke risk is unlikely to prevent Sapien from becoming “a new standard of care” for patients who don’t have other options, said Larry Biegelsen, an analyst with Wells Fargo & Co.’s Wachovia.
The risk of stroke could fall over time as surgeons perfect their implantation of Sapien, he said.
Next up for Edwards is a two-part randomized, controlled study for the latest version of the valve, the Sapien XT.
The latest version received European regulatory approval in March and Edwards now is selling it there.
The first part of the trial is set to look at up to 450 patients with severe, symptomatic aortic stenosis, according to Edwards.
The second part of the trial will compare traditional open-heart surgery patients with Sapien XT, which is delivered via a catheter threaded through the femoral artery in the thigh or through a small incision in the ribs.
That trial’s expected to take place sometime next year.