Rox Medical Inc., one of the county’s better funded medical device makers, wants to help people with chronic lung diseases breathe easier.
San Clemente-based Rox is working on a device to treat chronic obstructive pulmonary disease.
The disease is a combination of chronic forms of bronchitis and emphysema. It affects about 22 million Amer-icans and is the fourth-leading cause of death in the country.
Rox’s device, still under development, is called the AC1 System. The company could seek Food and Drug Admini-stration approval by 2015.
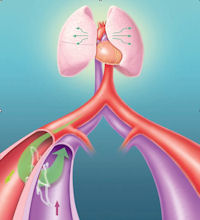
AC1 is an implant that creates and maintains a fistula, or a passageway, between an artery and a vein in the hip.
The passageway directs oxygen-rich blood from an artery to a vein, raising the amount of oxygen in the blood on its way back to the lungs, according to Rox.
The implant’s effects may include improved lung and heart function, such as reducing the resistance to blood flow within the lung’s blood vessels, the company said.
“You put a device into an artery and a vein, and the cascade of events that happens is more drug-like,” said Rodney Brenneman, Rox’s chief executive and cofounder.
AC1 is “additive” to current treatments for the disease, Brenneman said, such as oxygen machines, steroid inhalers and other drugs.
Rox has 25 workers, all but four of whom are in San Clemente. It assembles its devices at its headquarters.
Other medical device makers have stumbled in their efforts to commercialize an alternative to drugs for treating lung diseases.
Redwood City’s Emphasys Medical Inc. sold itself in 2009 after the FDA rejected its medical device that shifted air to the healthy parts of lungs in emphysema patients. Pulmonx Corp. of Palo Alto bought Emphasys.
“Until somebody gets through and has an approval, it’s hard to know how to measure success,” Brenneman said. “We are going to demonstrate success and define it.”
Rox is seeking to enter a market that Brenneman estimates is valued at $9.75 billion a year in the U.S. and Europe.
Rox plans to start selling its device in Europe later this year, Brenneman said. He projects the company’s yearly revenue to be in the “six figures.”
Started in 2004, Rox has raised nearly $50 million in venture capital.
Backers include several well-known healthcare venture capital names: Menlo Park-based Versant Venture Management LLC, which has a Newport Beach office; Essex Woodland Health Ventures of Palo Alto; Prism VentureWorks of Needham, Mass.; and Domain Associates LLC of New Jersey.
When Versant learned of Rox’s treatment, “We were very intrigued,” said William Link, a Newport Beach-based managing director for the firm.
“We feel it has great promise,” he said.
Rox received its last round of funding in 2008 for $35 million. It plans to raise more money in 2011, according to Brenneman.
U.S. sales are a few years off, Brenneman said, because the Food and Drug Admin-istration is so “variable.”
“We’d have to read the tea leaves on the FDA, which is, as you know, incredibly difficult these days,” he said.
Rox is expected to finish a pilot third-phase trial in the third quarter and then hopes to negotiate a pivotal trial, which would give the company data it could use in an approval application for its implant.
The company is open to an initial public offering or being bought, according to Brenneman.
A mechanical engineer by training, Brenneman previously worked for USGI Medical Inc., another San Clemente-based device maker.
He cofounded Rox with Keegan Harper, the company’s chairman and a serial healthcare entrepreneur. Rox is based on technology developed at Stanford University.
Several of Harper’s startups have Versant’s backing, such as implantable defibrillator maker Cameron Health Inc., also based in San Clemente.
Another Harper company, Ablation Frontiers Inc.—a maker of catheter-based medical devices for treating irregular heartbeats—was bought for $225 million by Minnesota-based device maker Medtronic Inc. last year.
Ablation Frontiers was based in Carlsbad.