Clinical-stage biotech company jCyte Inc. is developing its cell therapy treatment jCell for retinitis pigmentosa (RP), a rare eye disease that causes blindness and has currently no FDA-approved treatments.
Last month, the Newport Beach-based firm announced the enrollment of the first patients in a Phase 2 study testing the efficacy of the therapy in improving vision and quality of life in patients with retinitis pigmentosa over a 6-month period.
“JCyte has the potential to help restore vision in patients with RP, regardless of genetic subtype, and we look forward to sharing updates as the study progresses,” John Sholar, chief executive of jCyte, said in a statement.
The company’s founders have ties to UCI Gavin Herbert Eye Institute, which was established by Allergan co-founder Gavin Herbert.
It was the first academic facility of its type in Orange County and has become one of the nation’s biggest ophthalmology research institutes.
All trial participants will be treated at the institute, according to the company.
The trial, expected to have approximately 60 participants once fully enrolled, concludes in 2026.
Disease Affecting 2M People Worldwide
Retinitis pigmentosa is a rare, genetic eye disease that typically begins in childhood with many patients becoming legally blind by the age of 40.
It causes progressive vision loss by destroying the rod and cone photoreceptors, nerve cells that detect light and color, in the retina.
Two million people worldwide are estimated to suffer from the disease, including approximately 100,000 in the U.S. This makes the disease the leading cause of
inheritable blindness, according to the company.
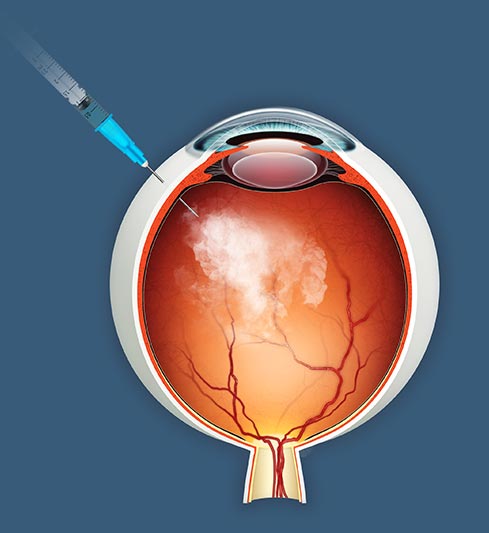
The company’s stem cell therapy, jCell, is a minimally invasive injection made directly into the back of the eye. It works by releasing growth proteins to reduce photoreceptor cell death and promote function of surviving photoreceptors.
The therapy aims to preserve vision by intervening when photoreceptors can be protected and potentially reactivated, according to the company’s website. In 2020, the company entered into a licensing agreement valued at $252 million with Santen Pharmaceutical Co., a Japanese pharmaceutical company valued at $3.6 billion, to commercialize in Europe and Asia (Tokyo: 4536).
Under the deal, jCyte received $50 million in upfront cash, $12 million in convertible debt and $190 million in future milestone payments.
This adds to some $47 million in funding that the company had raised to date.
JCyte’s plans for commercialization in the U.S. or overseas haven’t been disclosed. Company officials declined to give a potential timeline.
UCI-Born Startup
JCyte was started by UCI professors Dr. Henry Klassen and Dr. Jing Yang.
In the fourth grade, Klassen experienced sunlight-induced headaches that led his physician to believe he might have retinitis pigmentosa.
While his headaches weren’t caused by the disease, the experience started a lifelong interest in the biology of the eye. He has since spent more than 30 years studying and developing treatments for retinal conditions with nearly 20 of those years spent as a professor of ophthalmology at the Herbert Institute and UCI School of Medicine.
Klassen learned about retinitis pigmentosa during graduate school, turning it into his life’s work.
He received $14.9 million in funding from the California Institute of Regenerative Medicine to study the disease and in 2012 co-founded jCyte with Yang based on this research.
Klassen and Yang initially set out to slow the rate of vision loss but were later encouraged by the FDA to explore using higher doses of its product to not only preserve but improve vision.
Currently, Klassen is chairman and Yang is chief scientific officer of the company.