Inari Medical Inc., which began in 2011, tried to prove for years it’s pioneering a new way of treating patients with blood clots in their veins.
Doctors nowadays are clearly impressed, having treated 8,600 patients this year, more than double the number of 2019 patients.
Wall Street loves the story, too, as the company’s stock (Nasdaq: NARI) has more than tripled since its initial public offering in May, with shares rising from $19 to almost $65 and a $3.2 billion market cap.
Analysts are projecting sales will more than double to $129.8 million this year and climb another 35% next year to $176 million. In 2018, its first year of sales, it reported $6.8 million.
Yet, Chief Executive Bill Hoffman isn’t quite satisfied with his product lineup.
“We’re expanding our R&D efforts pretty dramatically, adding a number of engineers, and we’re working on a whole slew of new products, not just in these disease states, but other venous disease states,” Hoffman told the Business Journal during a Nov. 30 tour of his Irvine headquarters that also includes manufacturing.
“We’re going to continue to develop world-class products, and lots of them.”
While Hoffman declined to quantify his future expenditures on research, saying he hasn’t yet told Wall Street, this year’s expenses are giving a clear direction.
Research and development has almost tripled this year to $11.9 million.
The company, which has plenty of cash on hand, $168 million as of Sept. 30, is hiring researchers and tripling the size of its headquarters with plans to move into a new 121,000 square feet facility elsewhere in Irvine.
“We like to tell our investors and our board—and our board is fully on board—that it’s not about making money, growth per se, or shareholder value,” Hoffman said. “All of those things are byproducts. It’s about treating as many of these patients as possible, because they are everywhere.”
Key Differentiator
Inari was founded by a group of executives experienced in medical devices. Co-founder Bob Rosenbluth served as CEO until Hoffman took over in 2015. Rosenbluth continued as chairman until last year when the position was taken over by Donald Milder, co-founder of Versant Venture Management LLC, which invests in medical device startups.
The company took a different product development approach than other medical manufacturers that made devices to take clots out of arteries, and then repositioned them to take them out of veins.
Inari’s engineers believed they should focus on the veins, where a catheter-based device allows physicians to take out large amounts of clots.
The ability to take out clots has “made a spectacular difference,” Hoffman said.
“There’s an emotional impact,” he said. “Physicians say it’s very satisfying. Patients get better on the table—all of a sudden, they can breathe again.”
Open Culture
Inari manufacturers, assembles and ships its devices from its Irvine headquarters. A work order generally takes 36 to 72 hours to turn around.
At its Irvine facility, manufacturing workers load custom-built machines with thin pieces of nitinol, which are braided together to create the mesh collection bags that catch and remove clots from the veins. Others sit next to each other inside of a cleanroom and assemble catheter components, while wearing masks, gloves and gowns. A filtration system turns over air inside of the cleanroom about 30 to 40 times per hour.
Inari has been built on a culture of openness, hyper-communication and transparency, Hoffman said.
That attitude extends to the physical, open office environment of its headquarters, where the only office-style rooms have been repurposed as conference rooms.
Sales professionals in the field often share patient stories and photos on a companywide Slack channel, detailing the cases of deep vein thrombosis (also known as DVT) and pulmonary embolism (known as PE) that can alter lives in an instant, and how Inari’s devices help patients.
For example, a healthy child with a broken leg or a regular person walking off a long flight can develop PE.
Inari shares these patient stories because it wants to demonstrate what Chief Medical Officer Thomas Tu described as a “light bulb moment, a magic moment.”
“Even some of the biggest naysayers, the people who say this isn’t needed—you take a look at that clot and you can’t say it doesn’t matter. It matters. It matters a lot,” Hoffman said.
Educational Push
Inari believes education is a critical component of moving its market forward.
“Physicians need best in class training, not just on our devices, but on the disease states,” Hoffman said.
Sometimes patients chalk up DVT to poor circulation, or physicians overlook the condition because these patients often suffer from chronic diseases such as cancer. Meanwhile PE is still commonly misdiagnosed as a heart attack, stroke or even pneumonia, he said.
Inari educates physicians through its Clot Warrior Academy videos, where Tu discusses new techniques and patient outcomes with other physicians. Sessions have been conducted virtually since April.
The company plans to build a state-of-the-art training center at its new headquarters, though it will continue to host virtual training sessions after the pandemic is over, Tu said.
“We’ve found it’s a way to reach more people more effectively,” he said, noting Inari has been able to gather 100 to 150 physicians in an online session, compared to 10 or 15 at an in-person event.
Live-streamed procedures are also on the docket for next year.
“There’s something compelling about seeing a doctor struggle with the day-to-day. It’s different from the prepackaged cases we’ve presented in the past,” Tu said.
The pandemic has also brought public awareness to the dangers of blood clots, which will benefit patients long after the coronavirus crisis passes, Hoffman said.
Innovations Ahead
Inari intends to make significant investments in clinical trials and product development in 2021, Hoffman said.
Initial results from Inari’s ‘Flash’ registry, which included 250 patients with intermediate- and high-risk PE treated with its FlowTriever device, showed only one death after 30 days.
By contrast, the national PERT Consortium Quality Database recently reported 30-day mortality rates for intermediate- and high-risk PE patients of 6.1% and 25.9%.
“Our goal is to design even more rigorous trials that can be published in leading medical journals,” Hoffman said. “We hope to change the standard of care simply by the existence of these data.”
Inari will also bring new devices and product enhancements to market in 2021.
The company’s FlowSaver device, which will allow physicians to reintroduce extracted blood to patients following clot removal, is expected to receive federal clearance in the coming months.
“We know that some physicians still hold back—don’t remove as much clot—because they’re worried about blood loss. This device will free physicians to do what they do best,” Tu said.
The FlowSaver, along with all the company’s PE accessories, will be offered to physicians at no additional cost.
Inari charges an average of $9,100 per procedure, according to the company’s annual report.
The company has stated it expects its gross margins to decrease from around 90% during the year to about 80% as a result of this promise to its customers.
“We don’t want physicians to have to make choices between getting the best possible outcome for patients versus the best economic outcome for the hospital,” said Hoffman. “Physicians should never be in that position and they routinely are.”
New HQ
Inari said in October it inked a new lease for nearly 121,000 square feet in the Spectrum area of the city, at Irvine Co.’s Oak Canyon Business Center. It’ll be taking up most of the largest building at the complex, at 6001 Oak Canyon.
In addition to office space, the new facility will also be used for research and development, production, assembly, manufacturing, distribution, training and other related activities.
Inari needs more space; its manufacturing team is fast-approaching 100 employees split between two shifts, Paul Koehn, vice president of operations, told the Business Journal.
The company listed 15 open positions on its website at press time, with openings in engineering, medical affairs and other areas.
Inari increased its workforce to about 300 in OC and 430 companywide in 2020.
The company declined to disclose its hiring plans for next year, though it noted its salesforce would likely increase at it targets commercialization in Europe and other international markets.
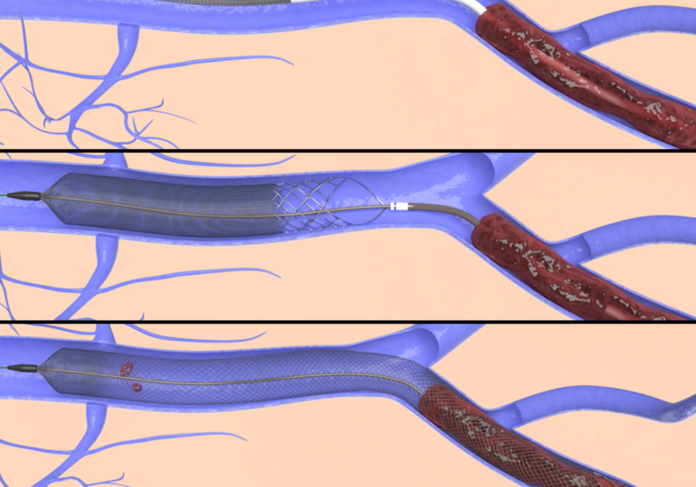
Inari’s Two Key Devices
Inari’s FlowTriever device treats pulmonary embolism, also known as PE, while its ClotTriever device treats deep vein thrombosis, or DVT.
“You’ve probably heard of other clot-removal devices, but you’ve never heard of devices that remove [this amount] of clot before,” Chief Medical Officer Thomas Tu told the Business Journal.
“It’s thousands of times the size of a clot that causes a stroke. That’s the fundamental difference.”
Inari estimates about 240,000 patients suffer from DVT, while another 200,000 patients develop PE every year. The company believes it has treated about 3% to 4% of its patient population.
DVT is a clot or series of clots that typically form in the legs and cause inflammation, pain and sometimes causes patients to lose their ability to walk. DVT can also lead to life-threatening PE.
PE occurs when a clot or series of clots travel to the lungs, which puts pressure on the heart. These patients often die as a result of heart problems.
“Think of putting your thumb over the end of a hose—that’s what this clot is,” CEO Bill Hoffman said. “It builds up pressure behind the thumb, and the heart can’t pump against the resistance.”
Historically, these patients have been treated with blood thinners because that’s what physicians are taught in medical school, said Tu, who left his own private practice to join Inari in mid-2019 after using its devices for several years.
Because PE occurs in the lungs but causes strain on the heart, different physicians such as cardiologists and vascular doctors took care of only pieces of it, but no one was leading the charge to improve treatment options and patient outcomes, he explained.
“In practice, you ask ‘Who’s the expert in this, who’s on top of the latest and greatest research?’ It turns out, there is no expert,” Tu said.