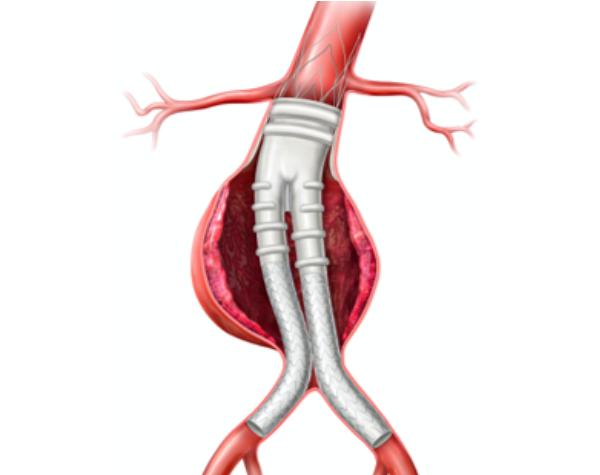
Focused on devices that treat aortic disorders, Endologix currently makes stents to treat abdominal aortic aneurysms, which occur when the lower portion of the body’s main artery becomes weakened. PQ Bypass’ first device is intended to address peripheral arterial disease in the lower limbs of the body.
Terms of the recent acquisition were not disclosed by Endologix.
News reports have placed the deal in the $300 million range, citing Richard Heuser, a cardiologist and entrepreneur who founded Milpitas-based PQ Bypass in 2009 with James Joye.
Such a price would mark the largest acquisition in the history of Endologix. It paid a reported $211 million in cash and stock for Santa Rosa-based competitor TriVascular Technologies Inc. in 2015.
“The acquisition of PQ Bypass is a seminal moment in Endologix’s history, building upon our leadership in the treatment of abdominal aortic aneurysm to champion disruptive technologies for the treatment of vascular disease,” Richard Mott, interim chief executive, said in a statement.
“Endologix doesn’t want to be another ‘me too’ company,” Chief Medical Officer Matt Thompson told the Business Journal. “We’re trying to be innovative—insurgents in the market. We’re hugely excited about acquiring PQ Bypass and [the deal] positions us beautifully for our goals in the future.”
2020 Restructuring
The acquisition of PQ Bypass caps a roller coaster year for Endologix, which was founded in 1992 under the name of Cardiovascular Dynamics, went public in 1996, and at one point counted a market valuation in excess of $1 billion.
The company, which operates out of a 129,000-square-foot facility in the Spectrum area of the city, saw its valuation fall well below that mark in recent years amid declining sales. Revenue steadily fell from $193 million in 2016 to $143 million in 2019.
The pandemic exacerbated the company’s issues, as it reported seeing a “significant reduction in procedural volumes” amid deferred hospital procedures. Revenue for the first half of 2020 was $53 million, a 25% drop from 2019 levels.
Endologix filed for Chapter 11 bankruptcy last July when it counted more than $260 million in long-term debt, which put a strain on its liquidity, according to court filings. After exploring a sale, the company ultimately agreed to be taken private through its largest creditor, Deerfield Partners.
The transaction completed in October, and allowed Endologix to shed $180 million in debt, including $130 million in debt held by Deerfield that was converted into equity. Endologix also gained $110.8 million in financing as part of the restructuring.
“We’re stable, we’re well-supported and we’re in a position to make acquisition and grow our business,” Thompson said last week.
Breakthrough Device
PQ Bypass makes the Detour system, which is designed to treat complex blockages in the femoral artery—or peripheral arterial disease (PAD).
PAD is caused by the buildup of fatty plaque in the arteries that reduces blood flow and can lead to muscle pain and weakness, sores and wounds, and, in severe cases, limb amputation. It impacts an estimated 8 million people age 40 and older in the U.S., according to the National Institutes of Health.
PQ Bypass was a perfect fit for Endologix because of “the combination of a clear, unmet clinical need, disruptive technology, and good clinical data,” Thompson said.
Data from a clinical trial showed 82% of patients didn’t experience major adverse events, and 83% of patients were clinically asymptomatic at two years post-procedure.
The company’s Detour system has received an expedited “Breakthrough Device Designation” status from the Food and Drug Administration and is expected to be ready for commercialization in 2022, Thompson said.
Global Launch
Endologix is already in the middle of a global launch for its newest stent graft.
The system, called Alto, is a minimally invasive treatment option for patients with abdominal aortic aneurysms, or AAA.
In the United States alone, an estimated 1.2 million people have an AAA, “many of whom will require a repair procedure,” according to the company.
The Alto system uses advanced sealing technology to protect healthy aortic tissue and provide long-term durability for patients, Thompson said.
“We firmly believe the durability of Alto is going to be superior to what it available on market today, and we plan to prove that,” Thompson said, noting this year the company will kick off a controlled randomized trial to track long-term durability of the Alto.
Durability is also an area of particular interest to FDA of late. The FDA announced last month it would hold an advisory panel in November to assess the kind of data it should be collecting, in addition to how to follow up with patients to determine long-term (five to ten year) durability of endovascular stent grafts.
Endologix in 2019 faced a class I recall of another of its abdominal stent grafts, the AFX, tied to 65 adverse events and five deaths. The FDA is currently advising anyone with an AFX stent graft to visit a doctor at least once a year to be checked for leaks.
The Future
After a turbulent 2020, Endologix is optimistic about its future.
The company is focused on developing aneurysm sealing technology as well as innovative tools that help surgeons “become confident and competent” with its lineup of products, according to Thompson.
For example, Endologix has already developed a virtual reality simulation for education and has live broadcasted several cases to demonstrate the Alto’s performance, he said.
Thompson said the company expects to hire depending on R&D and regulatory needs; it currently lists 13 open positions on its website.
A new chief executive could also be in store for Endologix this year.
Acting CEO Mott took the top spot from John Onopchenko during the going-private transaction last year. Mott was formerly chief executive of Kyphon, a publicly-traded medical device company that Medtronic acquired for $3.9 billion in late 2007.