Edwards Lifesciences Corp.’s traditional heart valves and critical-care products remain a steady beat of a company that’s seen a highly touted new device grab the spotlight.
Investor and analyst attention has been intently focused for the last year or so on the Irvine-based device maker’s new, less-invasive Edwards Sapien valve. It’s seen as the biggest development in heart valves in years, with the potential to open up the market to patients who aren’t candidates for major surgery.
The potential market for the less-invasive valves is expected to hit more than $1 billion in annual sales worldwide—and Edwards has a lead.
Yet Sapien remains a relatively small part of the company’s lineup for now, accounting for about 20% of the company’s $1.6 billion in revenue last year, when sales were largely confined to Europe. Sapien sales are expected to climb to about 30% this year, based on a recent approval for sales in the U.S.
Meanwhile, traditional heart valves—which require full surgery—brought in about $677 million for Edwards last year. Critical-care devices, such as blood monitors and catheters, saw sales of $508 million.
The two lines together accounted for 70% of the company’s revenue, and both are still growing. Traditional heart valve sales were up 7% last year, while critical-care devices notched a 12% gain.
Edwards is “pleased that there is continued adoption of the premium price and the premium products that we offer,” Chief Executive Michael Mussallem said at the company’s analyst day.
Traditional heart valves have roots in Edwards’ founding. They grew out of work done by electrical engineer Miles “Lowell” Edwards, who co-developed the Starr-Edwards mechanical heart valve in the early 1960s.
Edwards went through several iterations, including a period of time as the cardiovascular unit of Chicago-based diversified device maker Baxter International Inc. It was spun out as an independent company nearly 12 years ago.
Critical-care devices, which are used in various hospital units, also are a key part of Edwards’ history and future, according to Mussallem.
“Adaption of our advanced monitoring products is continuing, and we gained share both domestically and internationally in our legacy products that have a great history,” he said.
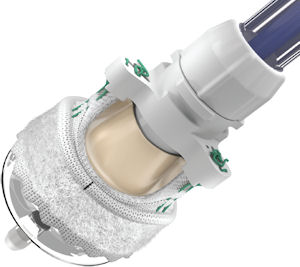
Edwards’ roster of traditional heart valves include the Carpentier-Edwards Perimount line, which are used to treat aortic and mitral valve disease.
The company last month received European regulatory clearance for a new heart valve called Edwards Intuity, which can be implanted through small incision surgery as well as traditional open-heart surgery.
The European clearance was for patients who have aortic stenosis, a “significantly undertreated” disease, said Donald Bobo, corporate vice president for heart valve therapy. “There (are) a lot of patients that are sidelined (who) really should come get therapy.”
Intuity provides “the ability to get all of the confidence and performance of our surgical valve platform, but in a procedure and product we believe is going to be much faster to implant (and) allows you to do small incision surgery,” he said.
Edwards now is discussing a possible clinical trial with the Food and Drug Administration for what’s called an investigational device regulatory application for Intuity, Bobo said.
That could help speed approval for sales in the U.S., although that’s likely “a few years away” in any case, he said.
The development of the Intuity has followed Edwards’ historical pattern of collaborating with doctors to develop devices. The company recently teamed with cardiologists, echocardiogram specialists and heart surgeons to implement valve clinics at hospitals, including a recently opened clinic at Hoag Memorial Hospital Presbyterian in Newport Beach.
Analyst Thom Gunderson of Minneapolis-based investment bank Piper Jaffray called the Intuity a potential catalyst in a research note issued after the company’s December analyst day.
“There is more to [Edwards] than Sapien,” Gunderson said.
GlucoClear
Gunderson also pointed to GlucoClear—a continuous blood-sugar monitor that Edwards co-developed with San Diego-based DexCom Inc. for hospital intensive care units and operating rooms —as another potential catalyst for critical-care product sales.
Edwards anticipates European regulatory clearance on the latest version of the device in the second half of 2012, according to the company.
“One reason we’re so good at (critical care) is that we’re very deep clinically and dedicated to this space,” said Carlyn Solomon, Edwards’ corporate vice president, critical care and vascular. “We’ve grown nicely over the past several years, and a lot of that is because of our strategy to innovate in our product lines.”
Edwards has brought out five new critical-care products over the past five years.
Offerings include FloTrac, a minimally invasive heart monitor; PediaSat, an oximetry catheter used on children; VolumeView, a sensor-catheter set, and EV1000, a monitor used to measure patients’ heart functions in surgical and intensive care settings.
Critical-care products, among other things, “give information that helps doctors and nurses determine if their patients are getting enough oxygen to their tissue. If you don’t get enough oxygen to the tissue, obviously, the tissue starts to die,” Solomon said.
They also are used to help fight sepsis, which occurs when the entire body is infected by bacteria. Sepsis can be fatal if untreated—it’s the 10th leading cause of death in the U.S., according to the Centers for Disease Control and Prevention.
Edwards sells its critical-care products to anesthesiologists and intensivists, who are doctors that work in intensive care units in hospitals and treat patients who have what Solomon called “very difficult cases.”
“Our products are used on the sickest of the sick, where life and death hangs in the balance,” he said.
Solomon said Edwards aims to educate clinicians on how the products save lives and hospital officials on how they save money.
Edwards primarily competes with smaller companies based in Europe on critical-care products. They face bigger, U.S.-based rivals such as Medtronic Inc. and St. Jude Medical Inc. on heart valves. Both are based in Minnesota and have significant operations in Orange County, with about 1,000 workers here between them.
Edwards is expecting steady growth for its traditional heart valves and critical-care products with a mix of ongoing research and development and acquisitions. The EV1000 and VolumeView grew out of the acquisition of a European company.
“We’re agnostic on where it comes from,” Solomon said. “We want the best ideas in our space.” n