Irvine drug developer Spectrum Pharmaceuticals Inc.’s stock is on the rise as it looks to new uses for its flagship drug and takes a step toward biotechnology.
Spectrum, which has annual sales of about $50 million, is seeking Food and Drug Administration approval for its Zevalin drug as a first-line of treatment for patients with non-Hodgkin’s lymphoma, a blood cancer.
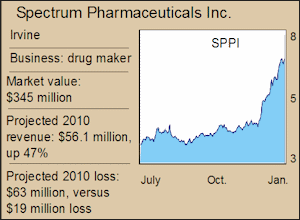
Wall Street expects Spectrum to post a loss of about $60 million for 2010, no surprise for a developing drug company.
And some analysts believe FDA approval for first-line treatment could turn Zevalin
into a blockbuster, an outlook that’s helped push Spectrum shares up 50% in the past 12 months.
The company had a market value of $345 million at recent check, with $92 million in cash on hand and “no long-term liabilities.”
Spectrum last month presented positive data from clinical trials showing that patients who received a Zevalin infusion were cancer free for a year. The company is expected to start a third-phase clinical trial this year and submit an application for FDA approval.
The drug currently is approved for treating the disease when other medications have failed or in conjunction with other cancer drugs.
Spectrum touts Zevalin’s ease of use as one reason to expand its reach to more patients.
“When somebody has cancer, the worst thing that happens to cancer patients is that the drugs that they get called chemotherapy makes them sometimes sicker than the disease itself,” Chief Executive Rajesh Shrotriya said.
Shrotriya quoted a researcher who said the “beauty of Zevalin” is that it’s given as a single dose, unlike many drugs used in chemotherapy, and doesn’t have “the usual toxicity” that’s commonly seen with those drugs.
“Would you rather take 16 courses of a drug or just one?” Shrotriya said.
Checkered History
Zevalin has had something of a checkered history. Spectrum is the fourth company to own it since it was approved in 2001.
The drug is proven to be clinically effective but historically had slow sales for several reasons, including the challenges of handling a radioactive drug, which could harm healthcare workers who administer it to patients.
Zevalin accounts for about 53% of Spectrum’s $55 million in annual sales.
“I think finally, after eight years of its approval and marketing, the stars are aligning for Zevalin because now, (there are) more and more patients (and) more doctors comfortable using the drug,” Shrotriya said.
The drug “will not disappoint,” said Justin Hall, a blogger on investor website Seeking Alpha, in a recent article about Spectrum.
“Needless to say, I think betting on Zevalin will continue to prove to be a smart move,” said Hall, who owns shares in Spectrum.
The FDA likely would be eager to approve Zevalin because it’s a one-time treatment that costs around $30,000, according to Hall.
That would enable the government to “dramatically cut the costs of treating (non-Hodgkin’s lymphoma) patients and namely those on Medicare,” he said.
Most of those patients currently are treated with a mixture of chemotherapy drugs and rituximab, a blockbuster antibody originally developed by Switzerland’s Roche AG.
Last week, Spectrum said it’s planning to develop what’s known as a “biosimilar” version of rituximab, which had sales of $6.1 billion in 2009. Rituximab also is used to treat rheumatoid arthritis.
Biosimilars are different from generic versions of drugs because no two biotech drugs can be exactly the same due to the sensitivity of materials to manufacturing processes.
Spectrum signed an agreement with Viropro Inc., a small drug maker based in Irvine, to develop the biosimilar of rituximab.
Roche sells rituximab as Rituxan and will lose patent protection in 2012.
Other Drugs
Spectrum also is seeking regulatory approval for wider usage of its Fusilev drug, which now is used to treat a rare form of bone cancer called osteosarcoma.
Fusilev, which was Spectrum’s first branded drug when it launched in 2008, is being considered as a treatment for colorectal cancer, a much larger market.
The FDA has told Spectrum that it will make a decision on that by April, according to Shrotriya.
Zevalin, Fusilev and two other drugs that are in Spectrum’s pipeline—belinostat, which treats lymphoma, and bladder cancer treatment apaziquone—“are the value drivers of the company,” Shrotriya said.
Spectrum’s working with Irvine drug maker Allergan Inc. to develop apaziquone.
Under terms of its deal with Allergan and others it’s working with, Spectrum could receive up to $450 million in payments based on meeting clinical, regulatory and commercialization milestones for apaziquone.
Spectrum is planning to submit an FDA application for apaziquone in 2012, Shrotriya said.
Shrotriya, whose career includes 18 years with New York’s Bristol-Myers Squibb Co., joined Spectrum in 2002 and oversaw its transformation from neurology to cancer drugs.
The company, which was started in 1987, once was known as NeoTherapeutics Inc.