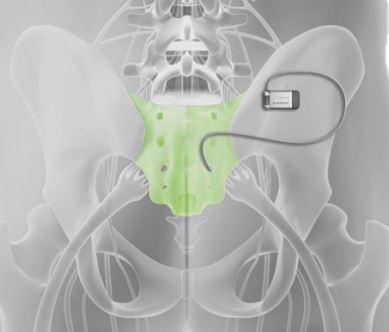
Device maker Axonics Inc. says its newest system designed to treat incontinence has demonstrated that the battery in the product can last more than two decades inside a body.
The University of Alberta implanted four patients earlier this month with the Axonics F15 and a stimulation showed “expected battery longevity in the body ranging from 18 years to over 22 years,” the Irvine-based company (Nasdaq: AXNX) reported in a Sept. 8 announcement.
The company’s sacral neuromodulation (SNM) system sends electrical signals to nerves in the spinal cord to help regulate the bladder and bowels. Axonics’ typical patient is a 55-year-old woman suffering from bladder incontinence.
The prior version of its main product had a battery life span of about 15 years.
“I am excited to now offer patients a recharge-free option that significantly increases device longevity in the body alongside the rechargeable Axonics SNM system that has helped so many of my patients,” Dr. Gary Gray, a urologist in the Department of Surgery at the University of Alberta, said in the Axonics statement.
Busy Summer
The latest device news isn’t all that’s providing a charge to Axonics, or its stock.
This summer has seen shares nearly double to $76 from a 52-week low of $38.41 in May, giving it a market value approaching $4 billion.
It’s close to its all-time high valuation; Axonics went public in November 2018 at $15 a share, raising $120 million.
In the past two months, the company has also announced:
The United States Patent and Trademark Office has granted eight patents and allowed four patent applications related to the company’s SNM technology in the past 12 months.
It raised $128.5 million in gross proceeds from an offering of 2 million shares priced at $63.85 each. Axonics said it would use the proceeds for working capital and general corporate purposes, including a potential $35 million milestone payment in connection with its prior acquisition of Contura Ltd., the maker of a hydrogel used to treat incontinence.
Second-quarter revenue rose 50% to $69 million, topping analysts’ consensus estimates. Its gross margin jumped to 72.8%, up from 62.6% in the same quarter a year ago.
“Q2 for Axonics was a beautiful quarter and things just kind of broke our way,” Chief Executive Raymond Cohen told analysts on a conference call.
“We’re just thrilled to death that we’re able to help so many people, who were suffering with this very annoying problem and to get those women dry.”
Ad Success
Cohen attributed the better-than-expected revenue to an advertising campaign called “Find Real Relief” that focused on women with any form of urinary incontinence.
“Our direct-to-consumer television advertising campaign, which we launched in April, is achieving our objective of increasing awareness for Axonics therapies, reducing stigma and encouraging women to consult physicians who specialize in bladder and bowel dysfunction,” Cohen said.
Overactive bladders affect an estimated 87 million adults in the U.S. and Europe, Axonics said. Another estimated 40 million adults are reported to suffer from fecal incontinence and accidental bowel leakage that the company’s product also treats.
Annual Forecast Boost
Cohen, who co-founded Axonics in 2013, has a long history in medical devices. Accolades include winning an Excellence in Entrepreneurship Award from the Business Journal in 2002, for his work with a prior company, Cardiac Science Inc., an Irvine defibrillator maker.
He previously told the Business Journal that Axonics’ launch of an implant to treat overactive bladders has had one of the three most successful debuts in American medical device history.
The company, which only began generating revenue in 2018, last month boosted its annual revenue forecast to $253 million, which is about a 40% increase from 2021 and up from a prior forecast of $238 million.
Axonics now employs nearly 530 people, including 230 in Irvine. It is Orange County’s 16th largest medical device maker by local headcount, according to Business Journal data.
Timing’s Everything
While the new F15 product is expected to work for decades, Axonics said a system from larger rival Medtronic has a battery that lasts only three to five years and must be surgically explanted to be replaced.
The Axonics F15 system, which was introduced earlier this year, utilizes a recharge-free implantable neurostimulator that is relatively small and thin at only 10 cubic centimeters in volume; it’s 20% smaller than its previous version, which ran about the size of a quarter.
The F15 has “taken off like a rocket ship, there’s no question about that,” Cohen told analysts. “The attractiveness of the F15 product where with a good implant, physicians can get over 20 years in a patient’s body, I think is something that no one has anticipated.”