San Clemente-based ReShape Medical Inc., which is developing an obesity-treatment device, has raised $15.8 million of a potential $22 million round of financing, according to a Securities and Exchange Commission filing.
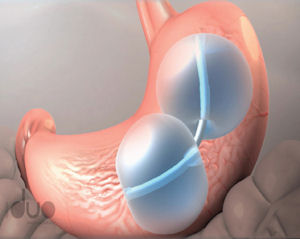
ReShape is developing a nonsurgical, dual-balloon medical device that occupies existing space in a person’s stomach in order to reduce its capacity for food. The ReShape Duo is designed to maximize the amount of space occupied in the stomach and conform to the organ’s natural anatomy for efficacy and stability.
Funding was provided by returning investors—Menlo Park-based venture firms New Leaf Venture Partners and U.S. Venture Partners and San Francisco-based SV Life Sciences.
Chief Executive Richard Thompson said the company aims to close on the additional amount in coming weeks.
“We are in the clinical stage and the financing is largely to cover the costs of the clinical trial,” Thompson told the Business Journal.
The company has an unconditionally approved investigational device exemption with the Food and Drug Administration and is “likely to be the first in our class to reach the U.S. market,” Thompson said.
Stomach
ReShape’s device is placed in the stomach during a 30-minute outpatient procedure using deep conscious sedation. The dual balloon, which can be inflated with saline, remains in a patient’s stomach for up to six months and is then removed.
ReShape Duo is approved in Europe and is being marketed for patients “who have exhausted their efforts with diet alone and wish to avoid or do not qualify for surgery.”
ReShape since 2007 has raised $40.3 million of financing including the newest round, according to its filing. That includes a $20 million round of funding in 2009.
The latest round is ReShape’s second in two months.
The company said raised $1.5 million in financing from unspecified investors last month.
Company Board
ReShape’s board includes several Orange County-area device veterans.
Chairman Kenneth Charhut is a former executive of Baxter International Inc.’s cardiovascular division, which eventually evolved into Edwards Lifesciences Corp. Board member George Wallace also worked at the Baxter cardiovascular operation and co-founded Micro Therapeutics Inc., an Irvine-based company that was bought by Ev3 Inc., which is now in turn part of Covidien PLC.
Robert Grant, another ReShape director, is chairman of Strathspey Crown LLC, a Newport Beach investment firm that has stated its intention to invest in “lifestyle medicine” businesses. Grant joined ReShape’s board in June.
Thompson said at the time of Grant’s appointment that “his expertise with cash-pay, elective medical procedures will be invaluable as we move toward commercialization.” The chief executive also pointed to Grant’s background as president of Allergan Medical, a unit of Irvine-based drug maker Allergan Inc. Allergan Medical’s products include the Lap-Band stomach band and the Orbera intragastric balloon, which is sold outside the U.S.
ReShape Duo “can be described as lifestyle medicine but it also bring real health benefits for the patients who need extra help in losing weight but are not interested in surgical procedures,” Thompson said.
San Clemente-based Intersect Partners, which ReShape director Wallace is also involved with, is another ReShape investor.