A total of 14 companies are racing to develop safe and effective COVID-19 vaccines and make 300 million doses available by January 2021 as part of the national program Operation Warp Speed.
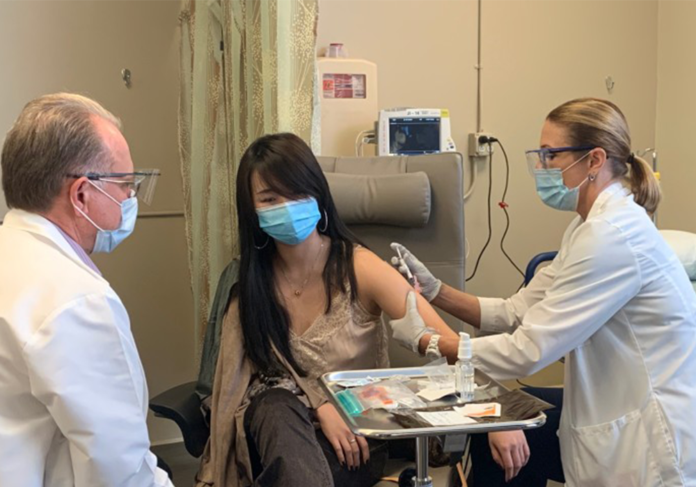
Hoag Memorial Hospital Presbyterian is playing a part in the efforts, as the site of a novel COVID-19 vaccine trial that kicked off late last month in Newport Beach.
The vaccine candidate, developed by a pair of Dr. Patrick Soon-Shiong’s biotech firms, contains two proteins that have the potential to produce long-lasting immunity to the virus, unlike the majority of other vaccines in development.
“It’s not only exciting, but inspiring and exhilarating,” Soon-Shiong told the Business Journal. “We’ve gone from having almost no knowledge of COVID to having a potential vaccine, one of the few that could stimulate both t-cells and antibodies for immunity that lasts longer, more than a few weeks or months.”
Soon-Shiong, the billionaire CEO of Culver City-based NantWorks LLC and owner of the LA Times, said his companies had experience working with Hoag on cancer trials and knew the hospital had early experience dealing with the coronavirus.
Hoag was selected as the first and only site to study the safety, side effects and immune response to the novel vaccine in a Phase I trial, which dosed its first patient on Oct. 20.
About 35 patients will enroll in the Phase I trial, and Phase II trials at three sites could start as early as December, according to Hoag officials.
Pooling Resources
Soon-Shiong started paying attention to the virus around the time of Kobe Bryant’s passing in late January.
The biotech entrepreneur, who owns a minority share in the Lakers and made his fortune by inventing the cancer drug Abraxane and subsequently selling his drug companies for nearly $9 billion combined, knew that his current enterprises under the umbrella of NantWorks could play a role in thwarting the coronavirus.
He said he recognized that a long-term vaccine would need proteins that deliver both antibodies that can block the entry of a virus and t-cells, which produce “memory” that trains the body to recognize specific antigens and triggers a faster, stronger immune response when it encounters the same antigen.
Soon-Shiong said he pulled together teams with cell manufacturing expertise from NantKwest, a cancer drug developer, and genomic sequencing expertise from immunotherapy developer ImmunityBio to work on the vaccine.
“We were in a unique position to harness the collective skill sets, brains, enthusiasm and scientific strengths to make this vaccine,” said Soon-Shiong.
Finding Hoag
NantKwest and ImmunityBio started working with Hoag Hospital on a Phase 2 pancreatic cancer cell therapy trial in August.
When the coronavirus pandemic hit, “Hoag quickly established itself as a center of excellence for COVID-19,” said Soon-Shiong.
He pointed out that Philip Robinson, medical director of infection prevention at Hoag, who is the principal investigator of the vaccine trial, treated the first patient with COVID-19 in the state of California in late January.
“It was Jan. 25 at 9:30 pm. I still remember,” Robert Braithwaite, chief executive of Hoag, told the Business Journal. “It was the beginning of something very momentous, and it has served the community well in the long run.”
It was the third diagnosed case in the U.S.
“I’m grateful our physicians didn’t relax,” Braithwaite said, noting Hoag doctors started setting up a delivery system in late January and February.
Early preparedness helped Hoag gain access to the experimental therapeutic Remdesivir, which allowed its teams to become “very competent and confident in treatment regiments,” Braithwaite said, and speed the subsequent distribution process for patients.
Remdesivir became the first drug to receive full Food and Drug Administration clearance on Oct. 22.
Research Infrastructure
The Hoag Center for Research and Education conducts more than 150 clinical trials every year.
While the center sees about 500 to 700 patients in an average year, it has enrolled 4,392 patients this year, with a large portion of patients participating in COVID-19 treatment and antibody detection trials.
Those numbers are “unusual” for a community hospital,” according to Clinical Research Director Deborah Fridman.
“We’re supported by the Hoag Hospital Foundation and philanthropy. Many community hospitals don’t receive the support that we do,” Fridman told the Business Journal.
“I’ve been given the go-ahead to do what we do best for the good of patients,” she said, noting support comes down from the hospital’s executive management team.
In addition to financial support, Hoag has the “unique capability” to quickly open a trial, said Fridman.
While an academic institution might take six months to a year to open enrollment, Hoag can start up a trial within four to six weeks.
Those advantages are possible because Hoag acts like a “priva-demic” hospital, rather than a traditional community hospital or an academic medical center, according to Braithwaite.
It prioritizes a robust research infrastructure to “give our community the opportunity to participate in leading-edge therapeutics, drugs and vaccines,” he said.
At the same time, the hospital can move a lot faster than an academic medical center because “we have the nimbleness, agility and streamlined nature of a community hospital.”
“The speed at which Hoag can move and did move during the pandemic has been very impressive. I’m sure it’s one of the reasons we had access to early-stage clinical trials and it speaks to the collaborative nature between the medical staff and the hospital.”
Philanthropic Support
Since January, Hoag has received more than $6.5 million in donations for COVID-related efforts.
Funds helped the hospital obtain supplies and provide patients with access to Remdesivir through a clinical trial within 48 hours, among other activities, according to Fridman.
Initial donations came from Fountain Valley’s Kingston Technology, Activision Blizzard, the parent company of Irvine’s Blizzard Entertainment, and donor Richard Scudamore.
Community members George and Julia Argyros, Joan and Andy Fimiano, and Ron and Sandi Simon also contributed.