Edwards Lifesciences Corp., the most highly valued publicly traded company based in Orange County, in recent weeks reported good news—including a significant milestone—and some not-so-good news.
The Irvine-based company (NYSE: EW), valued at about $45 billion, said it received federal approval for its minimally invasive Sapien heart valves to expand into the market for “low-risk” patients—those who typically receive open-heart surgeries.
Shortly thereafter, it also revealed that the delivery system for some of its heart valves, the Sapien 3 Ultra, were classified by the government as being under recall.
The heart valve itself is not part of the recall, only the delivery system, the company said.
Low-Risk Use
The Food and Drug Administration on Aug. 16 gave the go-ahead for the use of Edwards’ Sapien 3 and the Sapien 3 Ultra catheters to treat aortic stenosis in low-risk patients after reviewing results from the Partner 3 study.
The Partner 3 trial study showed the Sapien 3 “experienced extraordinary outcomes” in these patients, said Dr. Martin B. Leon, director of the Center for Interventional Vascular Therapy at New York-Presbyterian/ Columbia University Medical Center.
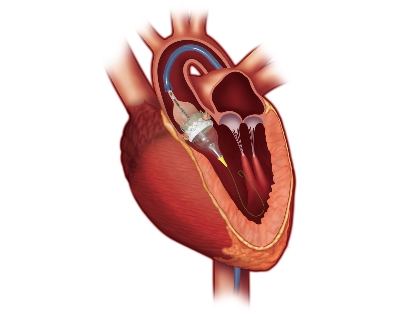
Edwards’ Sapien family of transcatheter aortic heart valves, known for its use in TAVRs, have treated hundreds of thousands of patients worldwide since 2007, when it was first commercially approved in Europe.
Previously, the valves were primarily reserved for the category of “high risk,” the very sick for whom traditional open-heart surgery is not optimal.
By contrast, low-risk patients are typically candidates for open-heart surgery.
The TAVR procedure—which is when doctors insert a catheter into a patient’s leg or left ventricle to guide a valve into the heart—may one day replace open-heart surgeries, according to at least one analyst.
In June, Edwards praised the U.S. Centers for Medicare and Medicaid Services for streamlining rules for TAVR, which may open the procedure to another new market—Medicare patients.
Preferred Treatment
The FDA ruling should make Sapien 3 “the preferred treatment” for patients with low-risk, severe aortic stenosis patients, according to Leon, who was the national co-principal investigator of an independently evaluated, randomized clinical trial that compared outcomes between TAVR and open-heart surgery.
Without treatment, severe aortic stenosis has a survival rate of 50% at two years, and 20% at five years post diagnosis.
About 13% of the population over age 75 has aortic stenosis.
Severe aortic stenosis “is a debilitating disease that often goes undiagnosed and is undertreated,” said Larry Wood, Edwards’ vice president, transcatheter aortic valve replacement.
“This approval is a significant milestone and will allow all patients diagnosed with severe AS to be considered for TAVR based on their individual preferences and anatomical considerations versus traditional risk scoring,” Wood said in a statement.
Edwards introduced the Sapien 3 in 2015 and has spent the last several years working to improve it; the Sapien 3 Ultra is its latest version of the product and one that has a slimmer profile.
Recall
Edwards expects the Sapien 3 Ultra valve system to eventually replace the Sapien 3, Chief Executive Mike Mussallem said in a July statement about the company’s second-quarter results.
However, on Aug. 22, Edwards disclosed the delivery system for the product had been designated by the FDA as a Class I recall.
Seventeen complaints of balloon rupture had been reported to Edwards, including one death by complication.
The incidence of rupture accounted for a 1% or greater malfunction rate when the Sapien 3 Ultra valve delivery system was used.
For a medical device to be considered “safe” by the FDA, there cannot be more than 1 per 200 people with severe complications, which translates to 0.5%.
As a result, 1,585 delivery systems have since been recalled, an FDA announcement said.
The company told the Business Journal that while the FDA utilizes the term “recall,” it doesn’t actually mean removal of the system; it’s a designation given when a manufacturer undertakes a remedial action.
The system remains on the market, a company spokesperson said. Technically, the designation indicates that the FDA issued a safety notice to customers and physicians with more detailed instructions, according to the company.
The FDA defines a Class 1 designation recall as “a situation in which there is a reasonable probability that the use of, or exposure to, a violative product will cause serious adverse health consequences or death.”
Centera Halted
The recall follows another decision when Edwards said in July it would pull the plug on a new transcatheter heart valve it’s been developing called Centera.
The device was designed to treat severe symptomatic aortic stenosis patients who typically need aortic valve replacement and are at immediate risk of open-heart surgery. As recently as April, Mussallem said enrollment in a trial for Centera would be completed next year.
Mussallem in July told analysts that discontinuing Centera was a difficult decision. He said that while it performed well, it still had certain issues that would have been costly to resolve, including more trials.
“The way that valve delivers didn’t work as well in all anatomies as we would have liked,” he said. “So it would have required us to make some enhancements to the delivery system.”
Edwards spent $363 million on research and development in the first half of the year, a 22% increase from the same period in 2018.