CathWorks, an Aliso Viejo medical technology firm trying to revolutionize the treatment of coronary artery disease, is one step closer to breaking through one of the world’s most impenetrable markets for device makers: Japan.
CathWorks announced the Japanese Ministry of Health, Labour and Welfare approved reimbursement for the company’s FFRangio System, following the device’s approval for regulatory use from the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) in late 2019.
The approval means hospitals will be reimbursed by the government and insurance companies for utilizing the device.
“The Japan reimbursement approval is an important step to enable more physicians and patients to have access to this innovative technology,” Dr. Hiroyoshi Yokoi, president of Fukuoka Sanno Hospital and vice chairman, Japanese Association of Cardiovascular Intervention and Therapeutics, said in a statement.
Japan’s rigorous clearance of CathWorks’ system is a “significant and important milestone,” Chief Executive Ramin Mousavi told the Business Journal.
“Even more difficult than approval is to receive reimbursement,” said Mousavi, a nominee for the Business Journal’s Innovator of the Year Awards earlier this month. “Most companies don’t even pursue Japan, especially not at a startup level.”
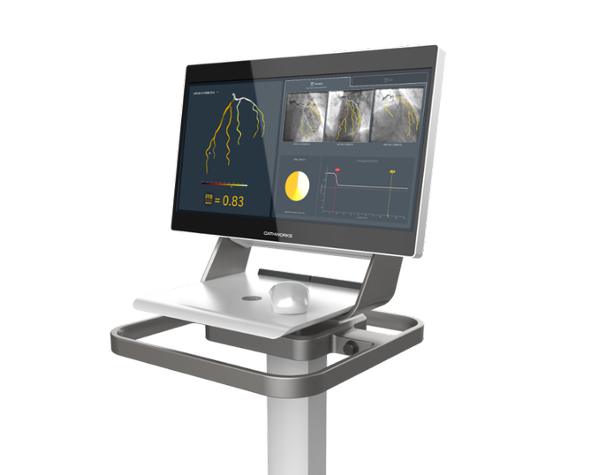
FFRangio System
The current method for examining coronary artery disease involves a process of using a dye system with catheters, anesthesia and medications.
CathWorks’ product takes a different approach by utilizing the 2D image of an angiogram to recreate a 3D color-coded image using proprietary AI technology.
It’s a noninvasive procedure that can transform X-ray images into color-coded 3D models of a patient’s heart in under five minutes.
“The FFRangio System enables us to quickly identify the physiologic significance of coronary artery disease (CAD) without the need for traditional pressure wires or hyperemic agents, which provides significant benefits to clinicians and patients,” Yokoi said.
CathWorks touts its procedure as saving $500 per patient.
The “FFR” stands for fractional flow reserve, which measures the level of blood flow in a diseased coronary artery, while the “angio” is short for angiography, or the examination of blood vessels by X-ray.
The company reported clinical trials have proven the FFRangio System to be not only noninvasive, but significantly more accurate at diagnosing coronary artery disease than traditional wire-based methods.
The company got FDA approval for the product in the U.S. about three years ago.
Mousavi, who took over the CEO role at the company this June, declined to provide current revenue. The company has 75 employees.
Japanese Adoption
This implementation could positively impact a significant portion of the patient population in Japan, where heart disease is the second-leading cause of death, and coronary heart disease is the cause of half of those deaths, the company said.
CathWorks aims to get its FFRangio System into Japan’s top 15 hospitals, including five by the year’s end.
“It’s an ambitious goal, but I’m confident we can get there,” Mousavi said.
The company earlier this year raised $30 million in a Series D round.
“I think our technology can transform the way coronary artery disease patients are diagnosed and treated,” Mousavi said. “It’s bold to change the landscape of this market, but I believe we have the technology to do it.”