R&D Medical Products Inc. has parlayed relationships and customer loyalty into growth.
The Lake Forest-based medical device contract manufacturer ranked No. 165 on the Business Journal’s annual list of fastest-growing private companies based in Orange County (see related stories throughout issue, list starting on page 50).
R&D’s revenue grew 15% to $4.2 million in the 12 months ended June 30 from $3.7 million in the 12 months ended on June 30, 2011.
R&D was founded in 1997 and has 32 workers. It manufactures and develops new medical devices for its clients.
“Our niche is when someone has [a device] that requires some type of electrode for either monitoring or stimulation,” said David Sheraton, R&D’s chief executive.
The company’s lineup also includes hydrogels, which are adhesives that can be used for wound care, cosmetic medicine and dermatology. R&D tends to develop products within niches.
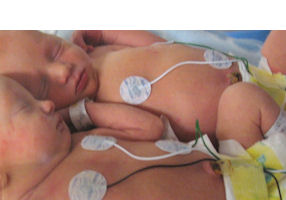
It worked on the Microlead electrode, which is used in neonatal intensive care units, or intensive care wards that care for infants who weigh 1.1 pounds or less at birth.
Sheraton said the company spent the first eight years of its existence developing devices for clients and helping them come to market.
“We knew it was going to be a slow growth initially,” he said. “We knew that at a certain point, once the development [and] regulatory cycle was finished, then things would start moving pretty quick.”
R&D has seen its business pick up in part because its clients’ products are now getting regulatory approvals and insurance reimbursement codes.
“Our growth is just beginning,” Sheraton said.
Clients
R&D has some large clients, although it mainly serves startups. Those include Medtronic Inc., which employs 700 people in a Santa Ana heart valve plant, and Valencia-based Neotech Products Inc., which makes devices used for neonatal intensive care.
VQ OrthoCare Inc., an Irvine-based maker of joint braces and other orthopedic support products, is another R&D client.
Sheraton said that the company’s clients typically come with a device concept in mind.
“It’s very routine that we’ll get a physician who wants to be an entrepreneur. So they’ll come to us initially with a proposed [device],” he said.
Loyalty
Sheraton said that loyalty is important, and he tends not to take on clients who have a similar concept to R&D’s existing clients.
“The ones I took to the dance, I want to continue dancing with,” he said. “Take care of our existing clients and maybe look at five new ones a year.”
R&D has a “very small” client base and does not do any marketing or advertising, Sheraton said.
The company turns away more than a few potential clients for strategic reasons.
“The development phase can suck up a lot of resources, and I’m very picky with my engineering team,” he said. “We like to get a really good feel with the client—is this going to be a viable entity?”
The company emphasizes its manufacturing and does have room to take on more of it.
R&D’s working on getting fully automated, “and when that client flips that switch, we’re going to be able to run with it,” Sheraton said, adding that the company wants to keep manufacturing in the U.S. and not outsource overseas.
R&D likes to be with its clients for a while. Sheraton noted that time for medical devices to get to market, including Food and Drug Administration and other regulatory approvals, runs from five to 15 years.