Vertos Medical Inc. set a goal for major expansion this year.
The provider of minimally invasive treatments for back pain is finding believers for its plans. On June 7, the Aliso Viejo-based company said it raised $26 million for commercialization and more research.
The Series C equity funding round, which the company says was oversubscribed, was led by new investor Norwest Venture Partners with participation from existing investors.
“The big difference for us was that we were proven commercially successful already,” Vertos Chief Executive Eric Wichems told the Business Journal.
“We doubled sales last year. There are still unpenetrated markets. The fact that we have a plan to get to profitability, sometime at the end of the year, attracted investors,” he added.
$130M+
Norwest, which manages more than $12.5 billion in capital, invests in early- to late-stage businesses across a wide range of sectors with a focus on consumer, enterprise and healthcare.
Since its inception, Norwest has invested in more than 650 companies and currently partners with more than 200 companies in its venture and growth equity portfolio.
Dr. Zack Scott, a general partner at Norwest, is joining Vertos’ board of directors. Scott’s specialty has been to help scale medical technology companies such as Cianna Medical, Coherex Medical and Evidation Health, among others.
“Lumbar spinal stenosis impacts 1 in 5 Americans over the age of 60 and Vertos’ procedure can help restore mobility and greatly improve the quality of life for these patients,” Scott said in a statement.
“Vertos directly aligns with our goal of partnering with companies that are focused on making a meaningful impact on patient outcomes and improving the healthcare system for all stakeholders.”
Vertos previously raised about $130 million in equity financing and another $27 million in convertible and non-convertible debt from investors that include Aweida Venture Partners, Leerink Revelation Partners, Mercury Fund, Onset Ventures and Pitango Venture Capital.
Cancer Beginnings
Vertos was founded in 2005 by a pair of interventional radiologists who designed a procedure to remove lesions off cancer patients.
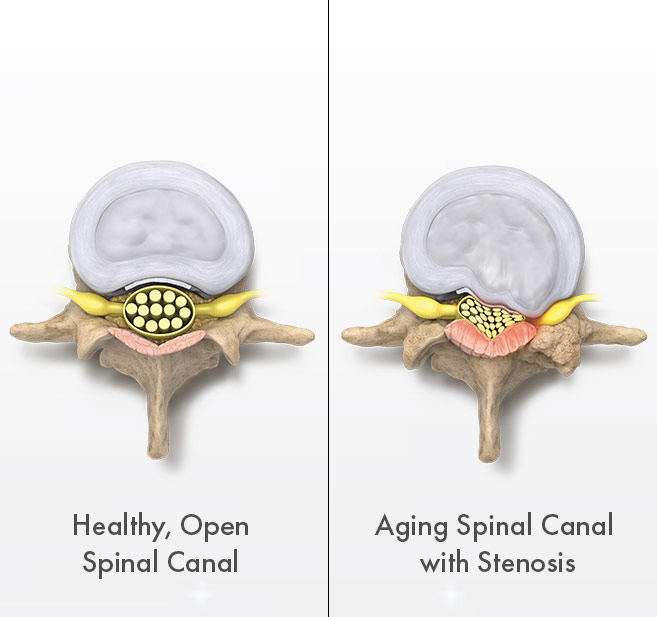
They realized the procedure could also be used to alleviate lumbar spinal stenosis, which limits the ability of people to stand and walk because of pain or numbness in their legs and lower backs.
People who suffer from it often find relief when sitting or leaning forward.
Typical lumbar spine surgery removes a layer of the vertebrae to create more space for the nerves.
Vertos’ procedure, which it describes as “mild,” is an image-guided outpatient procedure that removes a major root cause of lumbar spinal stenosis through an incision smaller than the size of baby aspirin.
The company said its technology “requires no implants, no general anesthesia, no stiches and no overnight hospital stay.”
Slow Start
The procedure received Food and Drug Administration clearance in 2006.
When it launched in 2008, there wasn’t a similar product on the market. However, in 2013, Vertos faced a setback when Medicare told the company that in order to receive reimbursements it had to conduct a significant clinical study, which caused a four-year delay in scaling the firm while the tests were conducted.
That action came about a year after a New York Times report cast doubt on the long-term success of the company’s procedures and its clinical trials.
The Medicare-requested studies have proven positive, as Vertos nowadays says it has 16 clinical trials over 30 peer-reviewed articles.
Vertos’ procedure has been clinically de-monstrated to last five years; patients typically return to activities of daily living within 24 hours with no restrictions.
The company says its safety profile is like an Epidural Steroid injection; its complication rate is less than 0.1% in all commercial cases.
“It actually has the safety of an injection but treats the symptoms like surgery does,” Wichems said.
Booming Sales
Wichems, whose background includes being director of marketing for medical devices in the plastic surgery market for Allergan Medical, joined Vertos in 2011 as its vice president of marketing before becoming CEO in 2014.
In recent years, growth has taken off. Last November, Deloitte Technology Fast 500 ranked Vertos No. 384 nationally, saying it grew 348% in a three-year period between 2018 and 2021.
Sales doubled in 2021 alone. It’s currently on a $60 million run rate with a goal of $100 million next year, Wichems said.
It backed off from an initial public offering last year because of market conditions; it may consider an IPO in a year or two, Wichems said.
“The path to profitability is putting us in a solid position,” he said.
In 2017, the OC tech and life sciences incubator Octane awarded Vertos its High-Tech Innovation Award.
At the time, the company only had 15 employees. Since then, it’s grown to 160 employees with plans for more than 200 by the end of this year.
R&D Plans
Some of the recently raised funds will go toward repurposing part of its offices to a research and development lab, Wichems said.
Last November, Vertos hired Mehrzad Khakpour as its first vice president of research and development. He was most recently at Sonendo Inc. (NYSE: SONX), a Laguna Hills-based dental technology company where he was responsible for developing the product portfolio and roadmap for the past 14 years.
Since his hiring, Khakpour has already built a team of 10 people.
When Vertos completes building its R&D lab, it will be maxed out on space, and will be looking for more, Wichems said.
70,000 Procedures
A few years ago, Vertos was doing 2,000 procedures a year; it’s now up to 6,000 a quarter with a goal of 30,000 a year. To date, more than 70,000 patients have been treated in the United States.
Wichems says as many as 2.4 million patients may benefit from the company’s back pain-treating procedure.
The company’s product can treat back pain much earlier than typical surgery and can be used on a larger pool of patients, he said.
About 95% of patients have five or more issues just on their backs alone. Future research might focus on knees because once patients receive injections for their backs, they can move about more often.
“There’s no such thing as having only one problem,” Wichems said. “Once you get the injection, you’re a lot more mobile.”