Irvine-based Edwards Lifesciences Corp. is back in court with a longtime rival as a critical regulatory hearing for its less-invasive replacement heart valve draws nearer.
Medtronic Inc. of Minneapolis filed two separate lawsuits against Edwards last month for alleged patent infringement on heart valves.
The suits come as the Food and Drug Administration’s circulatory systems device panel plans a July 20 meeting to discuss a pre-market approval application for Edwards Sapien.
The review is a key part of the approval process for using the device on patients who have severe aortic stenosis, or an abnormal narrowing of the heart’s aortic valve opening.
The FDA generally follows the advice of its regulatory panels on drug and medical device reviews.
U.S. Release
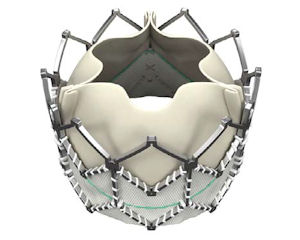
Edwards has said it could introduce Sapien in the U.S. later this year.
The valve is inserted into a patient via a catheter, eliminating the need for open-heart surgery. It and competing products, such as Medtronic’s CoreValve, are considered the biggest advancements in heart valves in years.
Sapien is expected to
be a game-changer for Edwards, which counts annual sales of $1.4 billion. The valve is approved in Europe and saw sales of $72.7 million there in the first quarter.
Analysts have pegged the market potential for less-invasive heart valves at more than $2 billion in yearly sales.
Edwards is expecting to spend some $40 million for the introduction of Sapien in the U.S. The company plans to have the valve available in 200 to 400 medical centers in the months following its introduction here.
Competition
Medtronic got its less-invasive CoreValve in a $700 million buy of Irvine-based CoreValve Inc. in 2009. It is said to be two years behind Edwards’ efforts to get Sapien on the market in the U.S.
CoreValve also is approved for sales in Europe. Medtronic doesn’t break out sales for the device.
It appears that Sapien will be approved to begin U.S. sales in the fourth quarter, said Larry Biegelsen, a medical device analyst with Wells Fargo Securities, part of San Francisco-based Wells Fargo & Co.
“We expect a positive panel recommendation and, ultimately, FDA approval of Sapien,” Biegelsen wrote in a client note earlier this year.
Suit Claims
One of Medtronic’s lawsuits claims Edwards infringed on its patent with Sapien.
Medtronic is looking for cash compensation for the unauthorized use of its technology, as well as a court order to prevent any future infringement.
Edwards doesn’t comment on pending litigation.
The other Medtronic suit contends that other Edwards’ products—such as the Perimount mitral replacement heart valve and the Embol-X Glide Protection System—use patented technology without permission, according to a Bloomberg news report.
Edwards and Medtronic have battled each other in courts in the U.S., Britain and Germany over Sapien and CoreValve patents.
Courts in Britain and Germany have found that CoreValve doesn’t infringe on Sapien patents.
Edwards won a $73.9 million verdict in the District Court in Delaware in February over similar allegations.
Medtronic is appealing that case.
The Delaware court rejected an additional request by Edwards to stop Medtronic from making and selling CoreValve. It also threw out Edwards’ claims for one of the patents in the lawsuit.
Edwards has said it would appeal those rulings.