An established Orange County medical device maker and a startup with a local manufacturing hub caught the attention of doctors and others last week with clinical trial results for less-invasive heart valves.
Irvine-based Edwards Lifesciences Corp. said that data from a clinical trial for its Sapien XT replacement aortic valve showed that death and stroke rates were similar to those for patients with an earlier version, the Edwards Sapien.
The data also showed that the new Sapien XT had a lower rate of other complications, including what the company called “vascular events” such as a perforation or hematoma after implantation.
Edwards was examining the Sapien XT in patients who cannot receive open-heart surgery, which is the traditional way to replace diseased heart valves.
“The Sapien XT was designed to reduce complications of the [transcatheter aortic valve replacement] procedure, and we believe this has been demonstrated by today’s result,” said Larry Wood, Edwards’ corporate vice president for transcatheter heart valves.
Edwards made the presentation at the American College of Cardiology’s annual scientific session in San Francisco.
Another company at the conference has OC links.
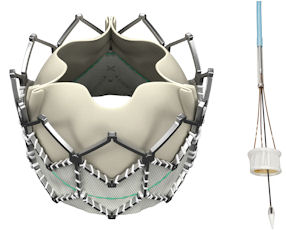
Santa Rosa-based Direct Flow Medical Inc., which manufactures its less-invasive heart valves in Lake Forest, said that patients who received its transcatheter aortic valve in its Discover clinical trial achieved “excellent survivability,” with better blood flow and minimal “aortic regurgitation”—where blood leaks back into the heart’s chambers—six months after implantation.
“When we talk about ‘excellent survivability,’ we have a 92% freedom from death at six months,” said Dr. Charles Davidson, an interventional cardiologist and Direct Flow’s chief medical officer.
Direct Flow has touted its valve design as something that prevents aortic regurgitation and other complications because it is flexible as opposed to an open, rigid metal structure, which is standard.
Davidson said that Direct Flow is going to continue its trial and will present 30-day outcome data at the European Association of Percutaneous Cardiovascular Interventions’ EuroPCR meeting, which is scheduled for May in Paris.
Transcatheter heart valves are designed to replace diseased heart valves in patients who have aortic stenosis, or a narrowing of the body’s main artery.
They are inserted with catheters through the leg’s femoral artery or through an incision between a patient’s ribs.
Edwards and Direct Flow are two of several companies pursuing the market, which is expected to be a multibillion-dollar opportunity in the coming years.
Other established companies include Medtronic Inc., which produces its less-invasive valves in OC and got into the field through a $700 million purchase of Irvine-based CoreValve Inc. four years ago.
Wall Street gave favorable reviews for Edwards’ trial results.
“Equally importantly, all-cause death and disabling stroke significantly declined when compared with the Partner I data, suggesting that the clinical benefits of Sapien may be better than originally anticipated,” Ben Andrew, an analyst with Chicago-based William Blair & Co., said in a research note published after the meeting.
Andrew wrote that while the Sapien XT data was “encouraging, we heard consistent feedback from clinicians that although newer implanting centers are ramping up steadily … there are several hurdles for these centers to overcome, which may take time to resolve.”
Edwards is planning to submit the data to the Food and Drug Administration as part of its effort to seek domestic marketing approval for Sapien XT. That valve is available overseas.
Analysts Brooks West and Michael Dinerman of Minneapolis-based Piper Jaffray said in a note that they expect “the new device should readily clear FDA approval hurdles and quickly become the standard of care” in transcatheter aortic valve replacement.
Direct Flow received European approval for its valve in January.
Davidson said the company has talked with the FDA about starting a domestic feasibility trial sometime within the next several months.
“We don’t have final approval yet, but we are in favorable discussions that make us optimistic,” he said.