Based in Irvine, device maker Axonics Inc. reported fourth-quarter revenue climbed 61% to $85.6 million to $86 million, topping the $78.1 million expected by analysts.
“This quarter’s record revenue result reflects the growing demand for our best-in-class incontinence products,” CEO Raymond Cohen said in a statement.
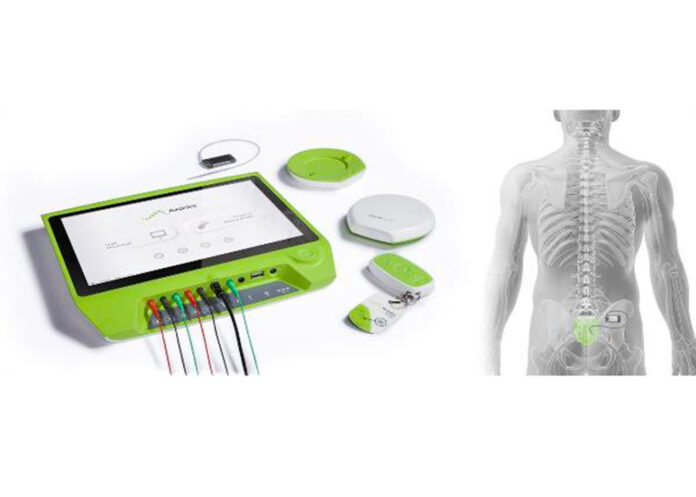
Axonics’ launch of an implant to treat overactive bladders has led to one of the three most successful debuts in American medical device history, according to Cohen, who has been a longtime executive in medical devices, including earning an Excellence in Entrepreneurship award from the Business Journal in 2002.
Axonics’ success was exemplified by a 4,509% growth in revenue over a two-year period, placing it first on the Business Journal’s list of Fastest-Growing Midsized Public Companies in 2021.
Its high growth should continue as the company forecast 2023 sales will climb 25% to $342 million, which also topped the average analyst estimate for $329.9 million.
In the trading session after the announcement, shares rose 4.8% to $64.73 and a $3.2 billion market cap.
Patients are becoming more aware of the company’s products and physicians are “overwhelmingly positive,” Cohen said. n
The company is awaiting FDA approval in the current quarter of its fourth-generation rechargeable sacral neuromodulation system. Currently, a patient needs to recharge the Axonics device for one hour every month. The fourth generation will reduce that to one hour every six months. Axonics expects the new version to receive FDA labeling saying its device will last 20 years in the body.