LoneStar Heart Inc. of Laguna Hills is taking a shot at treating advanced heart failure.
The developer of cardiac restorative therapies for patients with heart failure recently said it raised $20 million in equity financing.
Now it’s plunging ahead with clinical testing and regulatory clearance for Algisyl-LVR, its lead product.
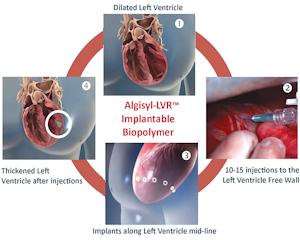
Algisyl-LVR is a single-use, self-gelling biopolymer that’s implanted into the heart’s left ventricle during surgery for treating advanced heart failure.
The National Institutes of Health estimates that nearly 6 million Americans are diagnosed with heart failure, with 550,000 new cases annually and 300,000 deaths from the disease.
Heart failure is primarily treated with drugs, with a few implantable devices available for use in later stages of the diseases, which is progressive.
Algisyl-LVR, which is a permanent implant, can be injected through what LoneStar President and Chief Operating Officer Frank Ahmann describes as a “window that is created by the rib cage.”
It’s a “literally straightforward injection from the outside of the heart into the heart wall, into the muscle that is diseased,” Ahmann said.
The company estimates that 4 million patients would be eligible for the device annually in the U.S. and Western Europe. It hopes to sell Algisyl-LVR primarily to cardiologists who treat more advanced stages of heart failure. Ahmann said the treatment will be hospital-based rather than done in a doctor’s office.
China-based Themes Investment Partners led LoneStar’s investment.
Milestones
Funding is contingent on LoneStar reaching certain milestones, Ahmann said.
Those milestones include getting a certain number of patients in its clinical trial, which is ongoing in Europe right now, and the transfer of the technology into a joint venture that LoneStar has established in China, he said.
The company wants to obtain European regulatory clearance in 2013, with U.S. clinical trials to follow.
FDA Approval
Ahmann said that “we may be about, realistically, two to three years away” from receiving Food and Drug Administration approval after LoneStar starts its domestic trials.
“The clinical trials are really designed to give us an answer about when this technology is eventually going to be effective in the treatment of congestive heart failure,” he said, adding that LoneStar’s early clinical work has focused mainly on safety rather than efficacy.
The company also is working on developing a way to bring Algisyl-LVR into the heart with a minimally invasive procedure.
“That’ll be available, hopefully in about nine months or so,” Ahmann said.
LoneStar has 10 workers and has raised $35 million since its founding in 2010.
Texas Ties
Its initial $15 million funding included several Texas-based equity investors.
The company has an office in Dallas and sponsors research at the University of Texas Southwestern Medical Center in Dallas and at the University of Texas, San Antonio.
LoneStar got its start with several technologies developed at the University of Texas.
Ahmann said LoneStar is a likely acquisition candidate in the future.
“As we’re all accustomed (to) here, the typical situation is that a large multinational company, either somebody like Edwards Lifesciences (Corp.), Boston Scientific (Corp.) or Medtronic (Inc.), end up acquiring the product, the technology and eventually do the marketing themselves,” Ahmann said.
Ahmann, a trilingual German passport holder who grew up in Mexico, is another device executive who comes from the Baxter International Inc.’s former cardiovascular unit family tree. Baxter spun the unit off 12 years ago as Edwards.
LoneStar’s chairman is veteran Orange County venture capitalist Olav Bergheim, who runs Fjord Ventures in Laguna Hills. Bergheim also worked at Edwards during its time as Baxter’s cardiovascular unit.
“There is a longstanding tradition of cardiac device companies here … we worked for some of them,” Ahmann said when asked why LoneStar was established in OC.
“This is where our private residence is; we don’t intend to move anywhere. We keep our families here,” he said.