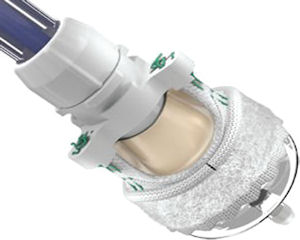
Irvine heart valve maker Edwards Lifesciences Corp. will start another domestic clinical trial in the months ahead.
The Food and Drug Administration gave the company a conditional exemption to allow a multicenter clinical trial for Edwards’ Intuity device, the first such U.S. trial of a “rapid deployment system for surgical aortic valve replacement procedures,” Edwards said.
The Edwards Intuity consists of a replacement heart valve made from cow tissue and a balloon-expandable frame. It’s designed to facilitate small-incision surgery and rapid valve deployment with a goal of enabling faster procedures.
Transform will study the safety and effectiveness of Edwards Intuity in a single-arm study that will follow standard heart valve guidance and use historical controls.
Edwards received European clearance for Edwards Intuity earlier this year.
Edwards also is being recognized for its less-invasive valves.
“We are encouraged by the company’s focus on developing its pipeline that should further strengthen its foothold in the [transcatheter aortic valve replacement] segment,” Zacks Investor Service said before the U.S. Intuity trial was approved.
Edwards recently also secured an FDA exemption for a trial involving GLX, a tissue treatment. GLX got European regulatory approval in May.
Zacks also said clinical trials for a pair of new transcatheter valves, Sapien 3 and Centera, are scheduled to start in Europe this year.

Complaint
Texas health officials are investigating a complaint centered on the marketing of a Fullerton-made medical device for weight loss without Food and Drug Administration approval, according to the Austin American-Statesman.
LipoTron 3000 is made by RevecoMED International Inc. of Fullerton and is distributed by Advanced Aesthetic Concepts of Fort Worth.
It uses radiofrequency electric fields that act like a microwave to heat the body.
A complaint filed with the Texas Department of State Health Services alleges Advanced Aesthetic was marketing the device for weight loss. The FDA has cleared LipoTron for medical uses, and doctors are allowed to use the devices for other purposes provided they don’t advertise it.
Surgery Gel
Abbott Medical Optics, a Santa Ana-based unit of Abbott Laboratories, continues to make progress on its push for new products.
The Food and Drug Administration this month approved an Abbott Medical product designed to protect eyes during cataract surgery. Healon EndoCoat coats the eye during cataract surgery and reduces damages to the eye.
Abbott said about 3 million people undergo cataract surgeries each year to replace blurry lenses in the eye. Abbott Medical, which was then known as Advanced Medical Optics Inc., got the Healon product line in 2004 when it bought the eye surgery business of Pfizer Inc.
Group Contract
San Clemente-based ICU Medical Inc. inked a pact with Resource Optimization & Innovation of Chesterfield, Mo., a St. Louis suburb, to distribute its MicroClave Clear neutral displacement needle-free intravenous connectors.
The connectors will be used on central and peripheral IV lines at Mercy, a Chesterfield-based organization that is one of the larger Catholic hospital operators in the U.S., serving more than 3 million patients a year. Resource Optimization is Mercy’s hospital buying group.
The MicroClave Clear combines ICU’s core Clave technology with a clear housing that lets doctors and other clinicians view the fluid path so they can clearly see whether they’ve completely flushed the connector after blood draws or administration.
Bits and Pieces
Atrilogy Solutions Group in Irvine said it was expanding its consulting services to help providers implement new diagnosis and procedure billing codes known as ICD-10. The Department of Health and Human Services has recommended that healthcare organizations be required to use the codes by October 2014. … Lake Forest home health provider Apria Healthcare Group Inc. said in a Securities and Exchange filing that Patrick Bourke III resigned from its board of directors, effective June 29. Apria said Bourke has not advised it of any disagreement with the company on any matter relating to operations, policies or practices. … Irvine-based Pacific Dental Services Inc. has agreed to buy 400 VELscope VX oral cancer screening devices over three years. The devices are made by Canada-based LED Medical Diagnostics Inc. and distributed by Henry Schein Inc. of Melville, N.Y. Financial terms weren’t disclosed.