Shares of enVVeno Medical Corp. plunged as much as 79% on Aug. 21 after it received bad news from the Food and Drug Administration.
The Irvine-based medical device maker on Aug. 20 announced that the FDA rejected the company’s Premarket Approval application for its lead product VenoValve, raising concerns about the product’s safety.
“We are obviously disappointed by the FDA’s decision,” Chief Executive Robert Berman said in a statement.
The FDA decision calls into question the future of a company that has the backing of two former top executives at Irvine-based Edwards Lifesciences Corp. Prior to the FDA announcement, enVVeno said it had enough cash, $35.1 million as of June 30, to last through the third quarter of 2026.
A day after the announcement, enVVeno’s stock sunk to 90 cents, the lowest price ever in the company’s seven years on Wall Street; at press time, shares traded at 92 cents with a $18 million market cap (Nasdaq: NVNO).
Berman said that the company will continue to work with the FDA on new criteria and “is assessing all options,” including a resubmission or appealing the FDA’s decision.
“We remain committed to the 2.5 to 3.5 million patients suffering from severe deep venous CVI in the U.S. and who have no effective treatment options and will continue to work with the FDA on new criteria to demonstrate the safety and effectiveness of our devices,” he said in the statement.
Company officials didn’t respond to the Business Journal’s requests for comment.
Edwards Ties
The company previously known as Hancock Jaffe Laboratories went public in 2018 where it raised less than $10 million.
It changed its name in 2021 to better reflect its products. EnVVeno has called itself “a company setting new standards of care for the treatment of deep venous disease.”
Its board of directors consists of Dr. Francis Duhay, the former chief medical officer at Edwards, and Dr. Sanjay Shrivastava, previously the principal research and development engineer for transcatheter heart valves at Edwards. Both have served on the board since 2018.
Last September, the company raised another $15 million in a public offering. Proceeds from the recent round went toward continued development for VenoValve and enVVe, according to the company.
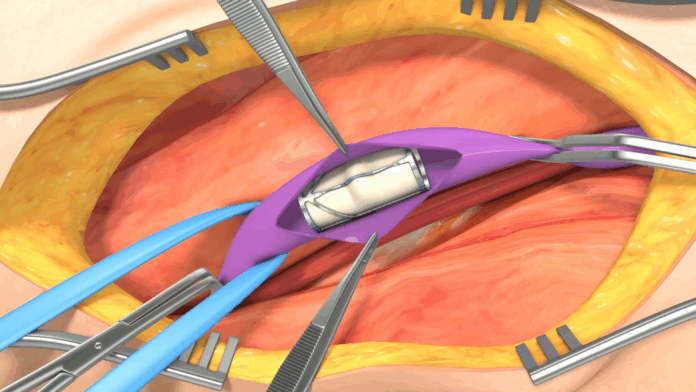
VenoValve is an implant that is in the late stages of development for the treatment of severe deep chronic venous insufficiency (CVI), a condition that occurs when veins in the leg get damaged, causing swelling, pain and in extreme cases, open sores known as venous ulcers.
The company is also developing a non-surgical, transcatheter-based replacement valve for CVI called enVVe, which the company is currently preparing to submit an investigational device exemption (IDE) application to the FDA.
Concerns Over Trial Data
The FDA questioned positive data from the company’s U.S. study, enVVeno said.
One-year data from the trial, which was presented last year, showed that 85% of patients experienced beneficial outcomes, including a reduction in ulcers.
EnVVeno measured these outcomes using the revised Venous Clinical Severity Score (rVCSS), a 10-item clinical assessment tool to evaluate and monitor chronic venous disease in patients. It defined clinical improvement as a three or more point improvement in rVCSS score, according to its website.
The FDA indicated that the data wasn’t sufficient on its own, and without blood flow measurements, improvements could have been attributed to bias or patients being enrolled in the study, enVVeno said.
“The results showed that a high percentage of the patients in the SAVVE study, who all previously failed standard of care treatments, showed significant clinical improvement after receiving the VenoValve,” Berman said.
“With the VenoValve being the only difference in their care, it is hard to not attribute the improvement to the VenoValve.”
Additionally, the FDA raised concerns about VenoValve as an open surgical procedure that required re-hospitalizations.
The company said it would “not expect to see similar safety events with a non-surgical replacement valve.”
Local Venous Disease Industry
The company said there are currently no effective treatments for deep CVI.
The current standard of care for CVI patients is elevating the leg or tightly binding it with compression stockings.
“We believe that venous disease has as much potential as arterial diseases did decades ago, to become a huge growth market,” Berman previously told the Business Journal.
VenoValve is surgically implanted via an incision in the upper thigh and inserted into the femoral vein, where blood flow is then restored.
EnVVeno is one of several local companies to focus on venous disease. One of them has been a clear success.
Irvine-based Inari Medical Inc., which designs medical devices to remove large blood clots from veins, was one of Orange County’s fastest-growing public companies, reporting revenue grew 63% over two years to $547.5 million for the year ended June 30, 2024. Last year, it was acquired for $4.9 billion by Stryker Corp.